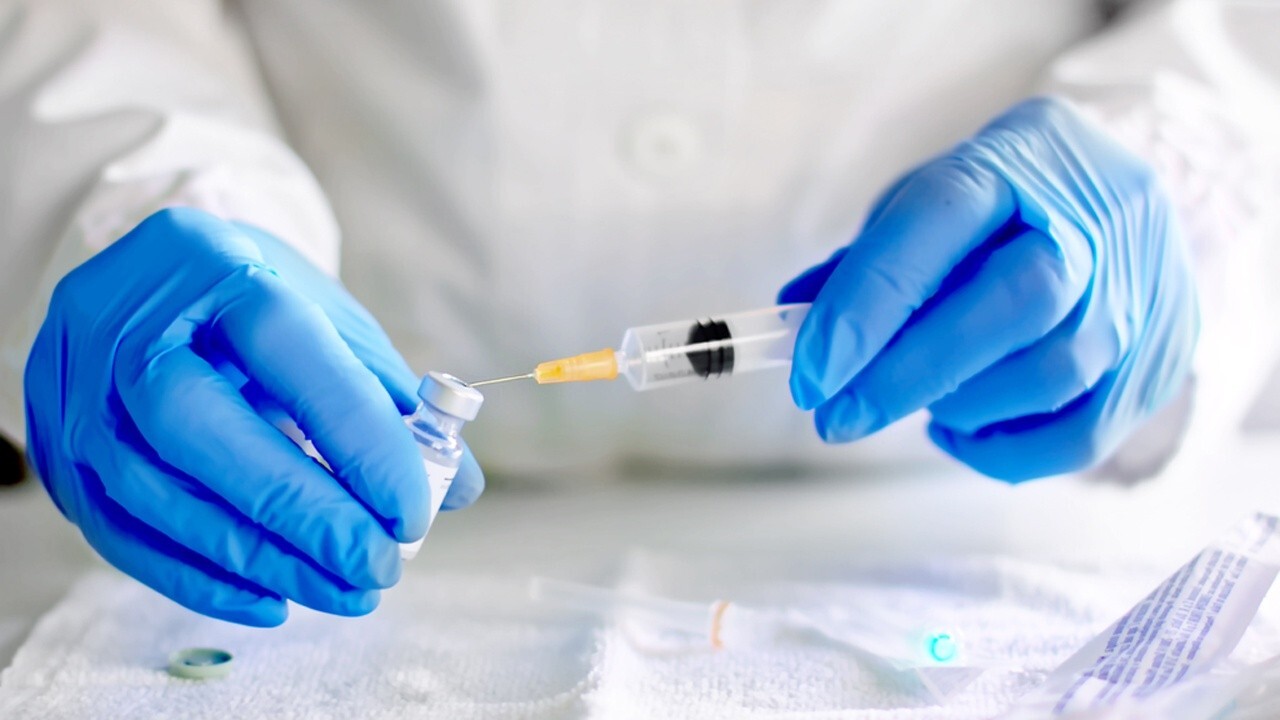
President Trump said Friday that the distribution of the Moderna vaccine will begin “immediately” after it was “overwhelmingly approved” this week, while signaling to European nations that have been “hit hard” that coronavirus vaccines will be “on the way” to they too.
“The Moderna vaccine has been overwhelmingly approved. Distribution will begin immediately,” Trump posted on Twitter.
CORONAVIRUS VACCINE SIGNING TRUMP EXECUTIVE ORDER AMERICAN PRIORITY FOR FOREIGN NATIONS
“Europe and other parts of the world are severely affected by the Chinese virus – especially Germany, France, Spain and Italy,” he continued. “Vaccines are on the way !!!”
The president’s tweet comes after the US Food and Drug Administration approved an emergency use permit (EUA) for the use of the Moderna vaccine on Thursday. However, the vote was not the final vote.
Despite the president’s tweet, FDA Commissioner Stephen Hahn said the FDA would “work quickly to finalize and issue an emergency use authorization.”
Hahn said the FDA also notified the U.S. Centers for Disease Control and Prevention and Operation Warp Speed so it could execute its vaccine distribution plans in a timely manner.
With regard to aid to European nations, the president last week signed an executive order ensuring that Americans have access to the coronavirus vaccine before the US government begins to help nations around the world.
Senior administration officials told Fox News last week that the president would reiterate to the American people that “the priority was an America First approach.”
“We work with the world,” Trump said last week when signing the order. “We work with the world.”
The president said the order ensures that “the US government gives priority to removing the vaccine to American citizens before sending it to other nations.”
A senior government official told Fox News last week that the “priority” is distribution to Americans and predicted that international assistance could come “in late spring, early summer” and after “vaccinating those [in the U.S.] who wish to be vaccinated “.
The FDA’s advisory board voted to approve the Modern coronavirus vaccine was 20-0, with one committee member abstaining.
The committee was asked to vote on the following question: “Based on all the available scientific evidence, do the benefits of the Moderna COVID-19 vaccine outweigh the risk of use in people aged 18 and over?”
TRUMP SAYS CORONAVIRUS VACCINES WILL “END PANDEMIC” MAKE PROGRESS IN “MIRACLE OF THE MODERN DAY”
Although significant, the committee’s vote in favor of the emergency use authorization (EUA) is not final. The vote will now go to FDA officials who will decide whether to grant EUA based on the committee’s findings. Such an approval will come about a week after the regulatory agency granted EUA to the Pfizer COVID-19 vaccine for people 16 and older, which states began launching to health care workers this week.
FDA review of emergency application for emergency use of Moderna coronavirus vaccine found no “specific safety concerns” in subgroup analyzes based on age, race, ethnicity, medical comorbidities, or previous SARS-CoV infection -2, which could pave the way for a second COVID-19 jab to enter the scene.
The group also found that the vaccine reduced the risk of confirmed COVID-19 – including severe cases – that occurred at least 14 days after the second dose.
FDA PANEL APPROVES MODERN VACCINE
The Committee carefully noted that there were insufficient data to assess the duration of vaccine protection, efficacy in certain high-risk populations, such as immunocompromised individuals, efficacy in persons previously infected with SARS-CoV-2, or efficacy in pediatric populations. There were also insufficient data for the committee to evaluate the future efficacy of the vaccine in relation to virus changes, efficacy against asymptomatic infection and efficacy against long-term effects of COVID-19, efficacy against mortality or efficacy against SARS transmission -CoV- 2.
Unlike the Pfizer and BioNTech vaccine, the Moderna cage caused non-serious side effects such as pain at the injection site, fatigue, headache, muscle aches, joint pain and chills. Reactions were generally characterized as mild to moderate.
CLICK HERE FOR FULL CORONAVIRUS COVERAGE
The FDA last week approved the Pfizer and BioNTech vaccine, which was distributed nationwide for healthcare and front-line workers this week.
In terms of affordability, the Moderna vaccine, which has been shown to be 90% more effective in late-stage clinical trials and requires two doses given weeks apart, does not require ultra-cold storage requirements and comes in smaller batches compared to those. 975 doses of Pfizer-BioNTech vaccine.
After the official approval of the FDA, which would mark the first such approval for Moderna, Americans could see a first 6 million doses distributed next week.
Madeline Farber, Alexandria Hein and Kayla Rivas of Fox News contributed to this report.