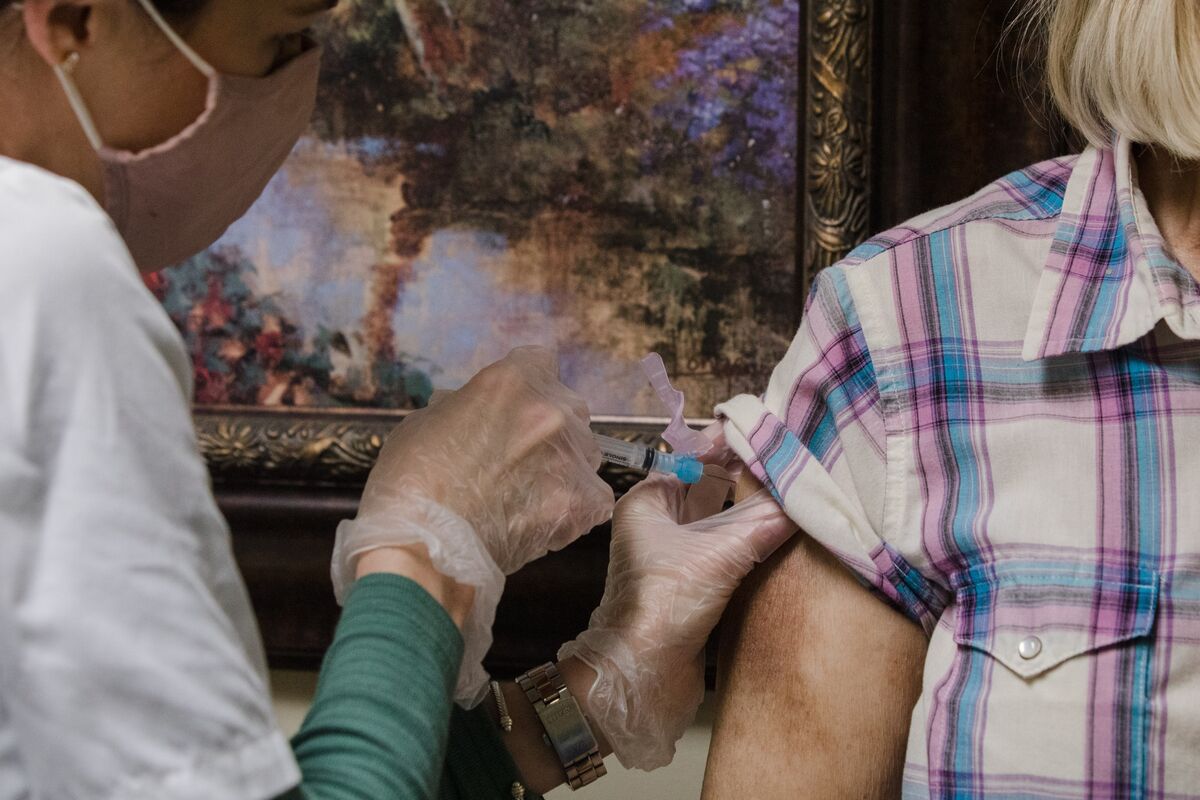
Johnson & Johnson’s one-shot Covid-19 vaccine cannot immediately ease the large-scale supply constraints that are slowing the US immunization campaign. However, it seems ready to help hard-to-reach populations that need protection against the virus.
This is the message of federal officials facing the best way to distribute J & J’s shot once regulatory clearance is expected in the coming weeks. The J&J vaccine, which can be stored in the refrigerator, offers a logistical advantage over Pfizer Inc. and Moderna Inc. vaccines with two injections currently in use, which must be kept frozen. However, it could take some time to change the pace of the campaign, a senior official said on Monday.
J&J has reached an agreement to supply the United States with 100 million doses of a single injection vaccine by the end of June. Andy Slavitt, Biden’s deputy pandemic response coordinator, said he would not be “too confident that these doses would come evenly.” Instead, Slavitt said he expects most of the supply to be available at the end of that period.
In a late-study study of more than 43,000 people, the J&J vaccine prevented 66% of moderate to severe cases of Covid-19, the company said Friday, showing lower levels of effectiveness than those developed by Pfizer, and Modern. But J & J’s shooting was particularly effective in stopping severe disease, preventing 85% of severe infections and 100% of hospitalizations and deaths.
Faced with the three safety and efficacy profiles of vaccines, dosing regimens and other requirements, US officials will establish a distribution model for them. Deliberating on the optimal use of each vaccine before adding another to the mix will be crucial, said Matthew Hepburn, the US government leader for the Covid-19 response.
The J&J vaccine is ready to provide faster protection than its counterparts for people in communities where the virus spreads rapidly, given that immunity increases immediately after a single shot, Hepburn said. This bodes well for places that seem vulnerable to us virus variants, he said.
The logistical ease of the J&J vaccine serves as a crucial public health tool, Hepburn said. “Honestly, it’s simply beautiful.”
The best movie
Anthony Fauci, the top U.S. infectious disease official, said Friday that federal authorities will discuss with governors, mayors and others involved in planning the immunization campaign to determine which vaccine should go where.
For example, in rural communities where authorities have challenged freezer requirements, J & J’s single dose may be more appropriate, Fauci said. If communities see fewer people returning for the second dose, a single vaccine could alleviate the problem, he added.
“There will be some local choice, depending on your needs,” Fauci said. “It’s a very pleasant problem.”
Public health experts want the federal authorities to make clear recommendations to states about which communities will benefit more from the J&J shot compared to other vaccines.
“States and localities should be given flexibility, but local decisions are better when made in the light of uniform national guidelines,” said David Fleming, chief physician and public health officer at PATH, a global health organization. non profit.
Fleming, a public health expert and epidemiologist who has held roles at the Centers for Disease Control and Prevention and The Bill & Melinda Gates Foundation has agreed that the J&J vaccine will be particularly useful in rural communities. He also suggested that it would be a good choice for people who are unable to travel to healthcare institutions and pharmacies.
The submission process
J&J received results from its large, late-stage clinical trial early last week. While the company has not yet provided details to show how the vaccine has worked in subpopulations, it said protection has been consistent across ages, races and regions.
J&J is now preparing documents for regulators in the US and elsewhere to apply for an emergency use permit. Scientific director Paul Stoffels said in an interview that he expects J&J to approach the US Food and Drug Administration by the end of the week and get a permit in March.
Once the FDA receives the J&J filing, it must convene a panel of external experts to review the data and make an independent recommendation. The CDC will hold its own meeting of external experts, known as the Advisory Committee on Immunization Practices, or ACIP, to provide guidance on how to allocate the vaccine and prioritize specific populations.
Together, these processes can take weeks. Pfizer and his partner BioNTech SE, for example, submitted an application for its vaccination schedule on November 20 and three weeks later was authorized for emergency use. Days later, the vaccine was first distributed.
Supply boost
To date, the United States has delivered 31.8 million doses of Pfizer-BioNTech and Moderna vaccines, according to Bloomberg’s Vaccine Tracker, with an average of 1.35 million doses per day given last week. Slavitt said there will probably be no “immediate dramatic change” when the J&J vaccine receives a green light from regulators.
Rather, the US will see an increase in supply by the end of the summer, he said. The government has reached agreements with Pfizer and Moderna for a total of 400 million doses by the end of the second quarter. With J & J’s additional offer, there should be enough photos to inoculate 300 million people by the end of June.
The former leader of Operation Warp Speed, the Trump administration’s effort to develop and distribute vaccines, said he expects the J&J vaccine to have a wider coverage than the other two vaccines so far.
“The fact that people will be vaccinated with a single shot is transformative,” Moncef Slaoui, a former chief scientific adviser to Warp Speed, said in an interview.
– With the assistance of Josh Wingrove